 
Evaporation of these acidic solutions affords the hydrate salt described above. A chromium(III) oxide coproduct is generated which is readily extracted into sulfuric acid. Anthroquinone and quinone are produced on large scale by treatment of anthracene and phenol with chromic acid. The most useful source of chromium(III) sulfate are the Cr(III) wastes from the chromate oxidation of various organic compounds. Other chromium(III) hydroxides have been reported. It results from the partial neutralization of the hexahydrates. 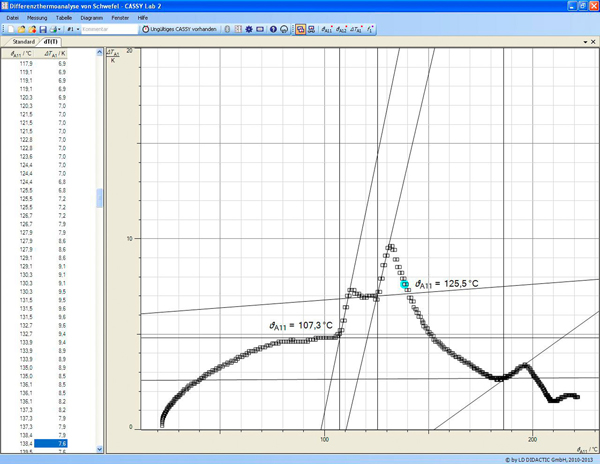
Most important commercially is basic chromium sulfate, which is thought to be SO4 (CAS#3). Further heating yields the anhydrous sulfate.Ī variety of other chromium(III) sulfates are known, but also contain hydroxide or oxide ligands. It is obtained by heating the 18-hydrate material above 70 ☌.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
